Ephedra
It took an unconscionably long time, but the federal government has finally managed to ban an unsafe dietary supplement before it can harm or kill any more unwary users - NYT
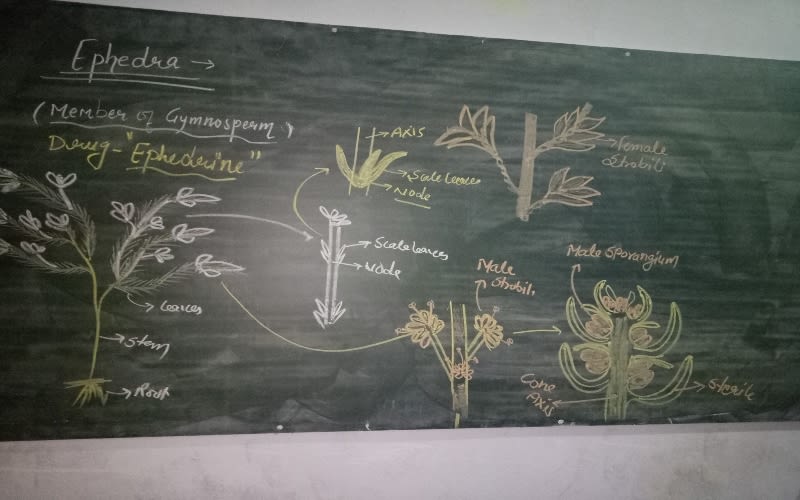
image by: Biology Blast
HWN Suggests
Stimulants in dietary supplements – when one is banned, another takes its place
In a study published last week, researchers at Harvard University examined 21 brands of dietary supplements containing an herbal stimulant called Acacia rigidula. More than half of the brands analyzed contained an untested amphetamine isomer called β-methylphenylethylamine (BMPEA).
The supplements were marketed to help people lose weight and to enhance energy. Even though BMPEA was first synthesized in the early 1930s, it is relatively unstudied. Because studies of safety and efficacy in humans have not been performed, its unknown whether it is dangerous. It appears that BMPEA is intentionally added to these supplements for its stimulant properties and since it is not listed on the…
Resources
 Ephedra Finale
Ephedra Finale
Health and Human Services Secretary Tommy G. Thompson announced that the Food and Drug Administration would soon outlaw U.S. sales of diet products containing stimulants derived from the Ephedra sinica plant.
After Ephedra, More Supplements Targeted for Ban
Encouraged by a Food and Drug Administration ruling last year banning weight-loss supplements containing ephedra, a consumer advocacy group targets at least a dozen other dietary supplements. The FDA had never before banned a dietary supplement, and it took years to agree on a ban on ephedra.
As Ephedra Fades, Consumers Snap Up Growing Alternatives
As ephedra steadily disappears from store shelves, a slew of new substances are angling to take its place as a top-selling weight-loss supplement.
Despite F.D.A. Ban, Ephedra Won't Go Away
Herbal medicine practitioners say they are relieved that their profession has not been included in the F.D.A. ban because ma huang is so useful.
Ephedra Is It Worth the Risk?
The herb ephedra, also known as ma huang, is a small, twiggy shrub native to Asia, where it has a long history of medicinal use, as documented in ancient medical treatises from India and China. Traditional Chinese and Indian medicine uses the branches of the herb to treat colds and wheezing and as a diuretic. Modern European practitioners of herbal medicine use ephedra only to treat symptoms of respiratory diseases (such as bronchial asthma).
Ephedra sinica
Ma Huang contains the sympathomimetics ephedrine and pseudoephedrine...
Risky stimulants turn up — again — in weight loss and workout supplements
The ingredients, apparently new, were popping up on the labels of dietary supplements marketed for weight loss and workouts. Sometimes the label said DMHA. Sometimes, Aconitum kusnezoffii. Or other, even harder-to-parse names.
Slim Pickings: Looking Beyond Ephedra
Ephedra, which has been used in China for centuries to treat asthma and other breathing problems, has been promoted in the United States for its ability to enhance sports performance and increase energy. Little evidence exists to support those claims.
The Ephedra Ban Is Not Enough
It took an unconscionably long time, but the federal government has finally managed to ban an unsafe dietary supplement before it can harm or kill any more unwary users.
 Stimulants in dietary supplements – when one is banned, another takes its place
Stimulants in dietary supplements – when one is banned, another takes its place
The story about synthetic stimulants popping up in dietary supplements is not new. A little over a decade ago the major dietary supplement of concern in the US was ephedrine (an alkaloid found in ephedra). Ephedrine was widely used for weight loss and performance enhancement (although it also has approved medical applications).
National Center for Complementary and Integrative Health
Ephedra is a low evergreen shrub with small scaly leaves. It has a long history of medicinal use in China and India to treat colds, fever, headaches, coughing, wheezing, and other conditions. The U.S. Food and Drug Administration (FDA) banned the sale of dietary supplements containing ephedrine alkaloids (stimulant compounds found in Ephedra sinica and some other plants) in the United States in 2004. Prior to the ban, ephedra was an ingredient in some dietary supplements promoted for weight loss, increased energy, and enhanced athletic performance.

Introducing Stitches!
Your Path to Meaningful Connections in the World of Health and Medicine
Connect, Collaborate, and Engage!
Coming Soon - Stitches, the innovative chat app from the creators of HWN. Join meaningful conversations on health and medical topics. Share text, images, and videos seamlessly. Connect directly within HWN's topic pages and articles.